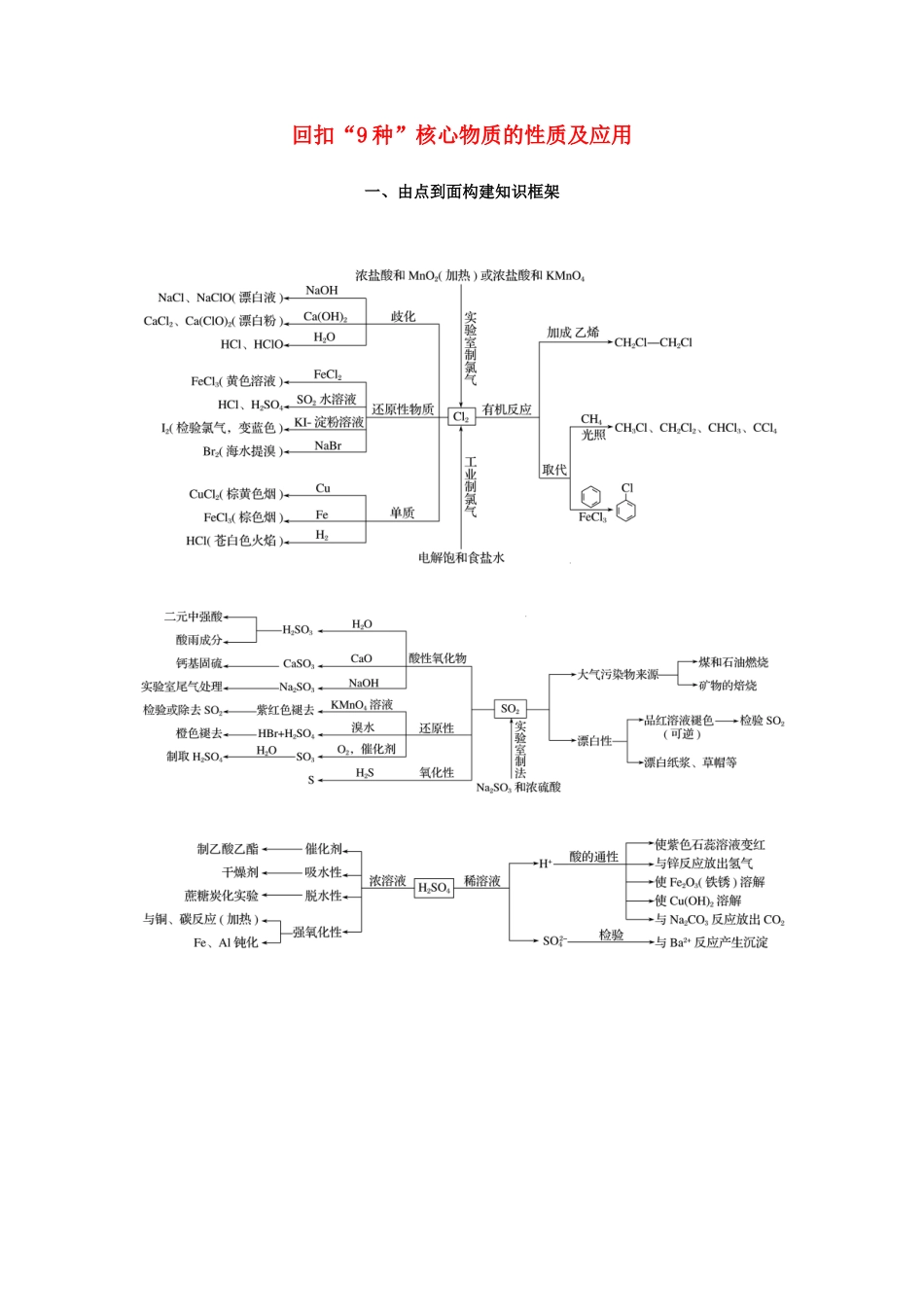
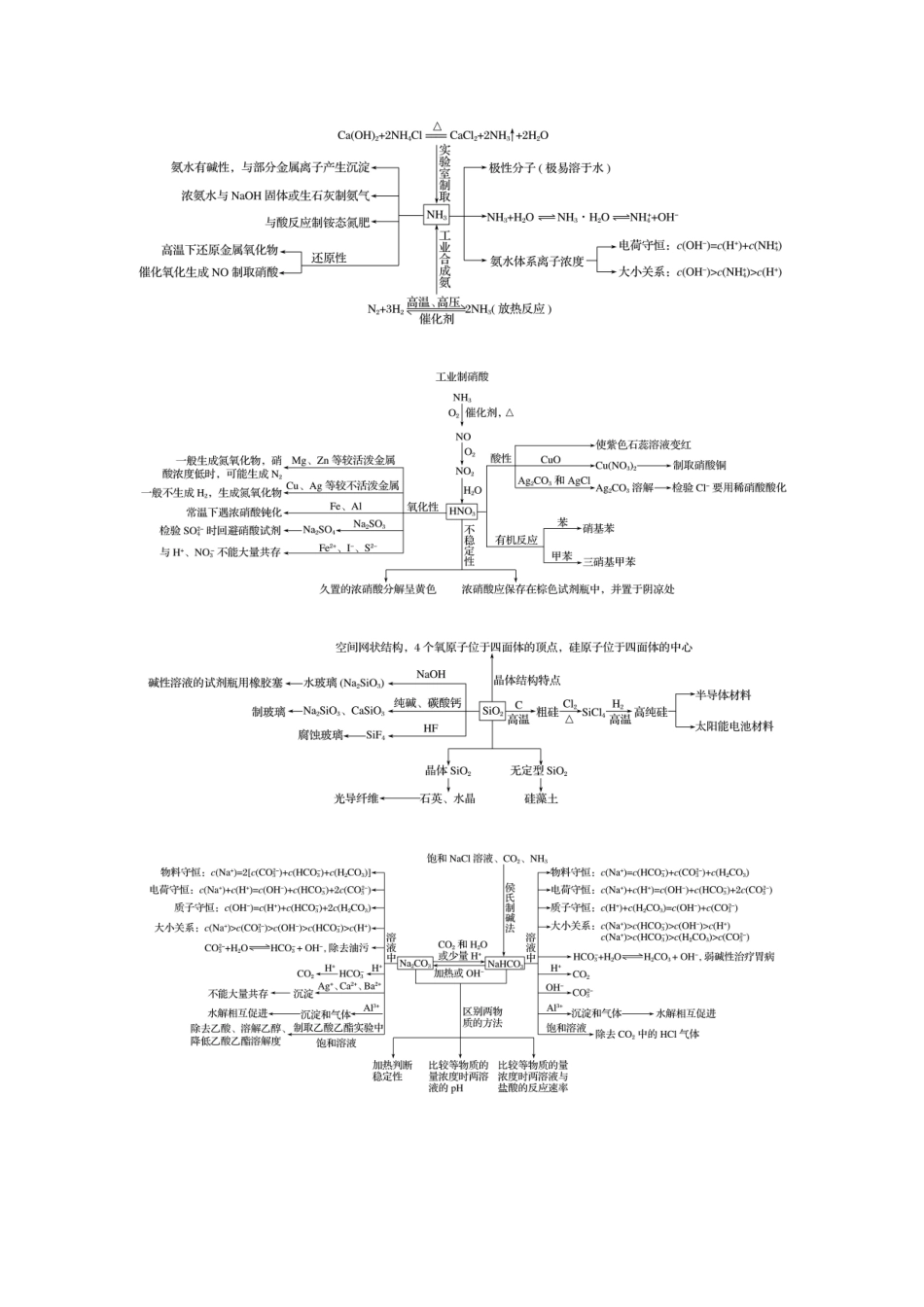
回扣“9种”核心物质的性质及应用一、由点到面构建知识框架二、重要方程式再现1.常见金属及其化合物(1)过氧化钠吸收空气中的二氧化碳:________________________________________________________________________。(2)过氧化钠溶于水:________________________________________________________________________。(3)纯碱溶液中通入CO2气体:________________________________________________________________________。(4)电解饱和食盐水:________________________________________________________________________。(5)铝片溶于烧碱溶液:________________________________________________________________________。(6)用AlCl3溶液和氨水制备Al(OH)3:________________________________________________________________________。(7)NaAlO2溶液与NaHCO3溶液混合:________________________________________________________________________。(8)泡沫灭火器的反应原理:________________________________________________________________________。(9)FeCl2溶液中滴加氯水:________________________________________________________________________。(10)用FeCl3溶液腐蚀铜片:________________________________________________________________________。(11)铝热反应(金属氧化物Fe3O4):________________________________________________________________________。(12)镁在二氧化碳中燃烧:________________________________________________________________________。答案(1)2Na2O2+2CO2===2Na2CO3+O2(2)2Na2O2+2H2O===4NaOH+O2↑(3)Na2CO3+CO2+H2O===2NaHCO3(或CO+CO2+H2O===2HCO)(4)2NaCl+2H2O=====Cl2↑+H2↑+2NaOH(或2Cl-+2H2O=====Cl2↑+H2↑+2OH-)(5)2Al+2NaOH+2H2O===2NaAlO2+3H2↑(或2Al+2OH-+2H2O===2AlO+3H2↑)(6)AlCl3+3NH3·H2O===Al(OH)3↓+3NH4Cl(或Al3++3NH3·H2O===Al(OH)3↓+3NH)(7)NaAlO2+NaHCO3+H2O===Al(OH)3↓+Na2CO3(8)Al3++3HCO===Al(OH)3↓+3CO2↑(9)2FeCl2+Cl2===2FeCl3(或2Fe2++Cl2===2Fe3++2Cl-)(10)2FeCl3+Cu===2FeCl2+CuCl2(或2Fe3++Cu===2Fe2++Cu2+)(11)3Fe3O4+8Al=====9Fe+4Al2O3(12)2Mg+CO2=====2MgO+C2.常见非金属及其化合物(1)实验室用MnO2和浓盐酸制氯气:________________________________________________________________________。(2)用烧碱溶液吸收尾气中的Cl2:________________________________________________________________________。(3)氯水滴入KBr溶液,比较Cl2和Br2的氧化性:________________________________________________________________________。(4)铜片溶于浓硫酸:________________________________________________________________________。(5)SO2使溴水褪色:________________________________________________________________________。(6)硫化氢与二氧化硫气体混合产生浅黄色沉淀:________________________________________________________________________。(7)SO2型酸雨在空气中放置酸性增强:________________________________________________________________________。(8)CuSO4溶液吸收H2S气体:________________________________________________________________________。(9)实验室用NH4Cl取制氨气:________________________________________________________________________。(10)氨气的催化氧化反应:________________________________________________________________________。(11)铜溶于稀硝酸:________________________________________________________________________。(12)久置浓硝酸变黄:________________________________________________________________________。(13)工业上制取粗硅:________________________________________________________________________。(14)硅酸钠溶液中通入少量CO2:________________________________________________________________________。答案(1)MnO2+4HCl(...