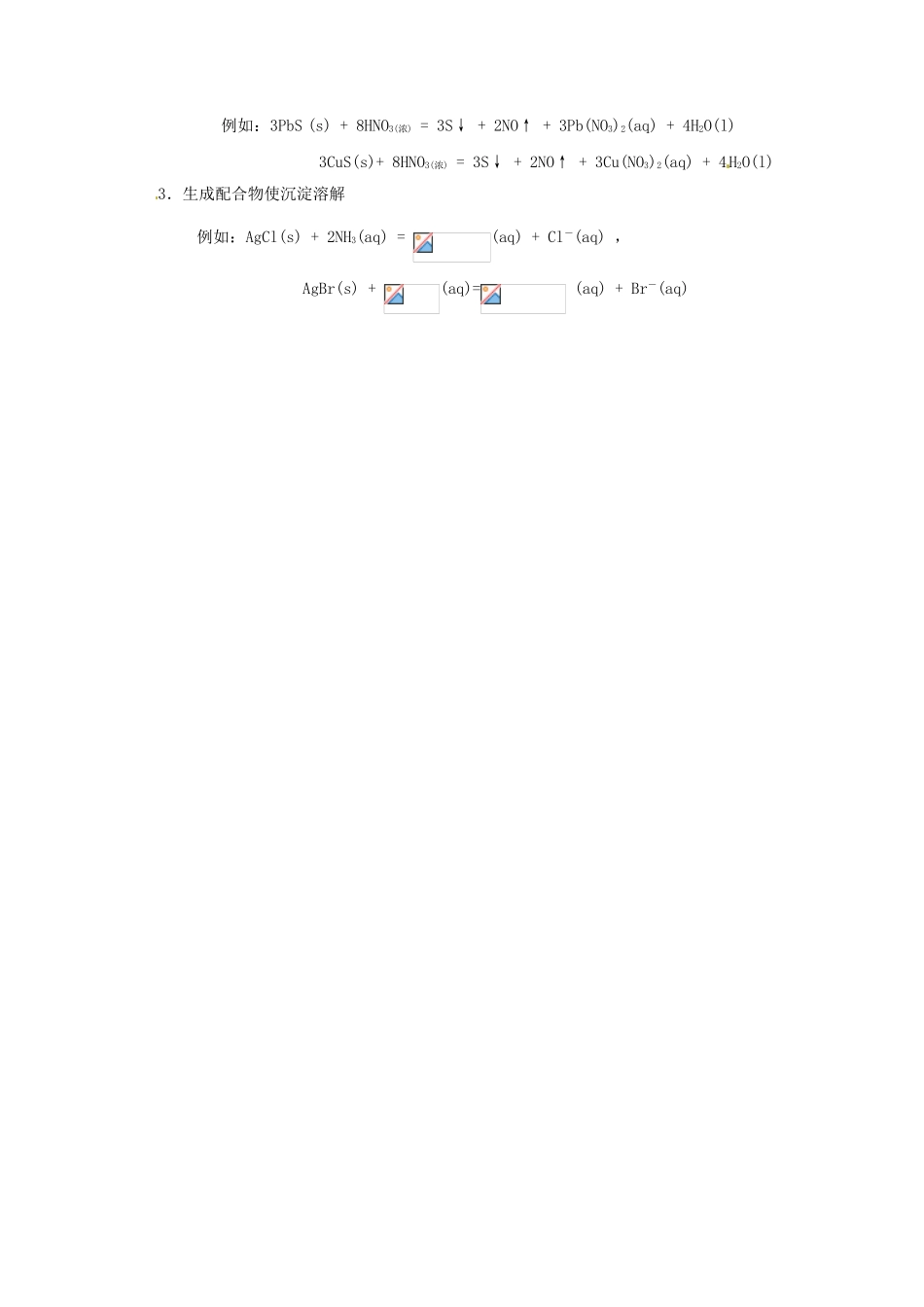
§5-2 沉淀—溶解平衡的移动Equilibrium Shift between Precipitation and Solution一、影响难溶电解质沉淀的因素(The Affect Factors on Precipitation of Undissolved Electrolytes) 1.同离子效应(Common –ion effect):难溶电解质的溶解度,因加入有共同离子的强电解质而降低的效应
Sample Exercise 1:Calculate the molar solubility of CaF2 at 25℃ in a solution containing (a) 0
0100 mol·dm3Ca(NO3)2(aq ) ; (b) 0
010 mol·dm3 NaF(aq)
Solution:设在(a)溶液中 CaF2(s)溶解度为 x mol·dm3
CaF2(s)CaF2+(aq) + 2F-(aq) 0
010 + x 2x (0
010 + x) · (2x)2 = 3
91011 , ∵ x