Impurities In New Drug Substances 新原料药中的杂质 Page 1 of 22 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE IMPURITIES IN NEW DRUG SUBSTANCES Q3A(R2) Current Step 4 version dated 25 October 2006 This Guideline has been developed by the appropriate ICH Expert Working Group and has been subject to consultation by the regulatory parties, in accordance with the ICH Process
At Step 4 of the Process the final draft is recommended for adoption to the regulatory bodies of the European Union, Japan and USA
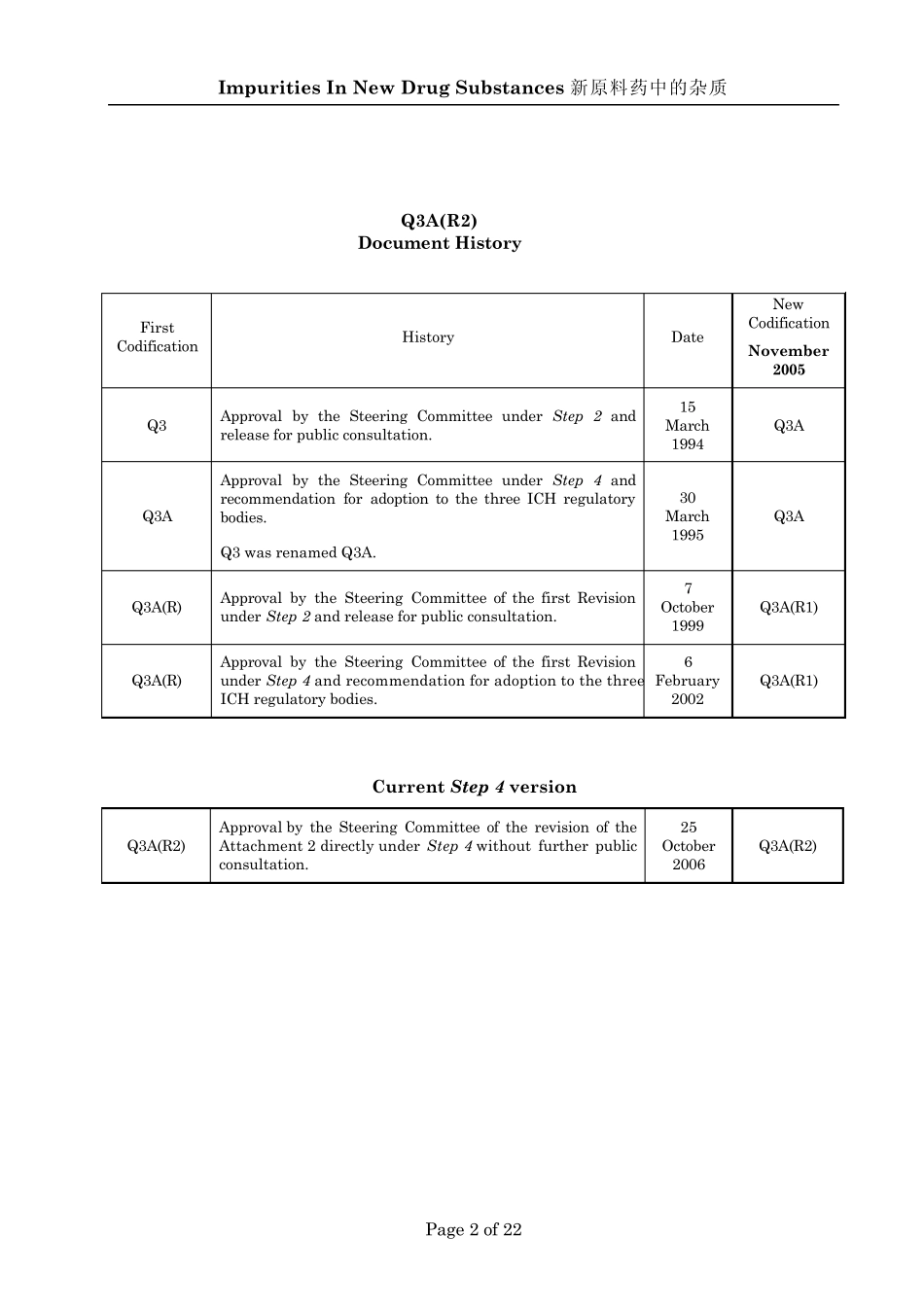
Impurities In New Drug Substances 新原料药中的杂质 Page 2 of 22 Q3A(R2) Document History First Codification History Date New Codification November 2005 Q3 Approval by the Steering Committee under Step