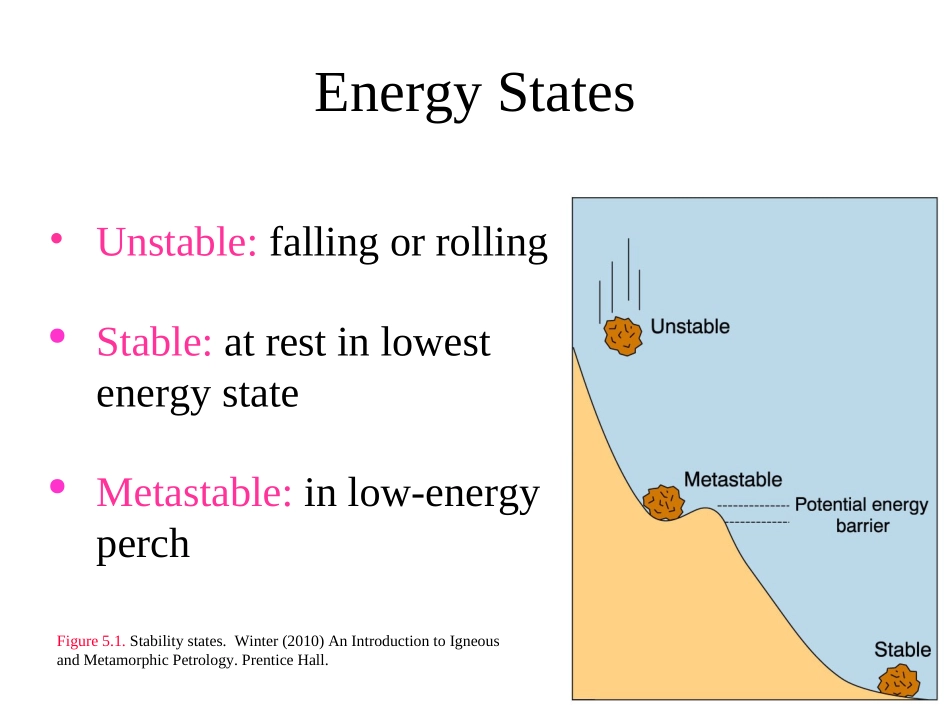
ThermodynamicsBeginwithabriefreviewofChapter5NaturalsystemstendtowardstatesofminimumenergyEnergyStates•Unstable:fallingorrollingStable:atrestinlowestenergystateMetastable:inlow-energyperchFigure5
Stabilitystates
Winter(2010)AnIntroductiontoIgneousandMetamorphicPetrology
PrenticeHall
GibbsFreeEnergyGibbsfreeenergyisameasureofchemicalenergyGibbsfreeenergyforaphase:G=H-TSWhere:G=GibbsFreeEnergyH=Enthalpy(heatcontent)T=TemperatureinKelvinsS=Entropy(canthinkofasrandomness)ThermodynamicsGforareactionofthetype:2A+3B=C+4DG=(nG)products-(nG)reactants=GC+4GD-2GA-3GBThesideofthereactionwithlowerGwillbemorestableThermodynamicsForothertemperaturesandpressureswecanusetheequation:dG=VdP-SdT(ignoringXfornow)whereV=volumeandS=entropy(bothmolar)WecanusethisequationtocalculateGforanyphaseatanyTandPb