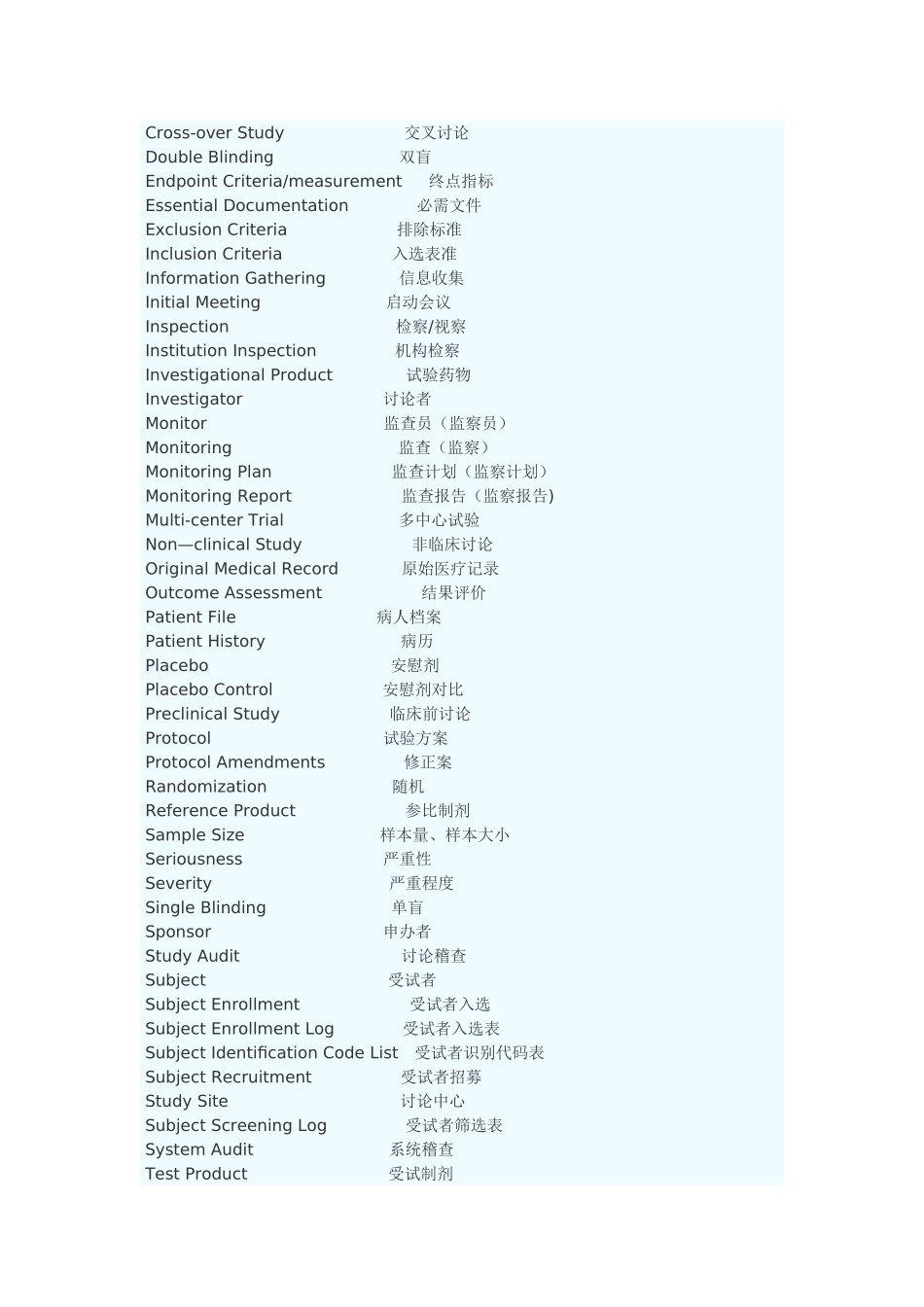
专业术语缩略语 英文全称 中文全称ADE Adverse Drug Event 药物不良事件ADR Adverse Drug Reaction 药物不良反应AE Adverse Event 不良事件AI Assistant Investigator 助理讨论者 BMI Body Mass Index 体质指数CI Co-investigator 合作讨论者COI Coordinating Investigator 协调讨论者CRA Clinical Research Associate 临床监查员(临床监察员)CRC Clinical Research Coordinator 临床讨论协调者CRF Case Report Form 病历报告表CRO Contract Research Organization 合同讨论组织CSA Clinical Study Application 临床讨论申请CTA Clinical Trial Application 临床试验申请CTX Clinical Trial Exemption 临床试验免责CTP Clinical Trial Protocol 临床试验方案CTR Clinical Trial Report 临床试验报告DSMB Data Safety and monitoring Board 数据安全及监控委员会EDC Electronic Data Capture 电子数据采集系统EDP Electronic Data Processing 电子数据处理系统FDA Food and Drug Administration 美国食品与药品管理局FR Final Report 总结报告GCP Good Clinical Practice 药物临床试验质量管理法律规范GLP Good Laboratory Practice 药物非