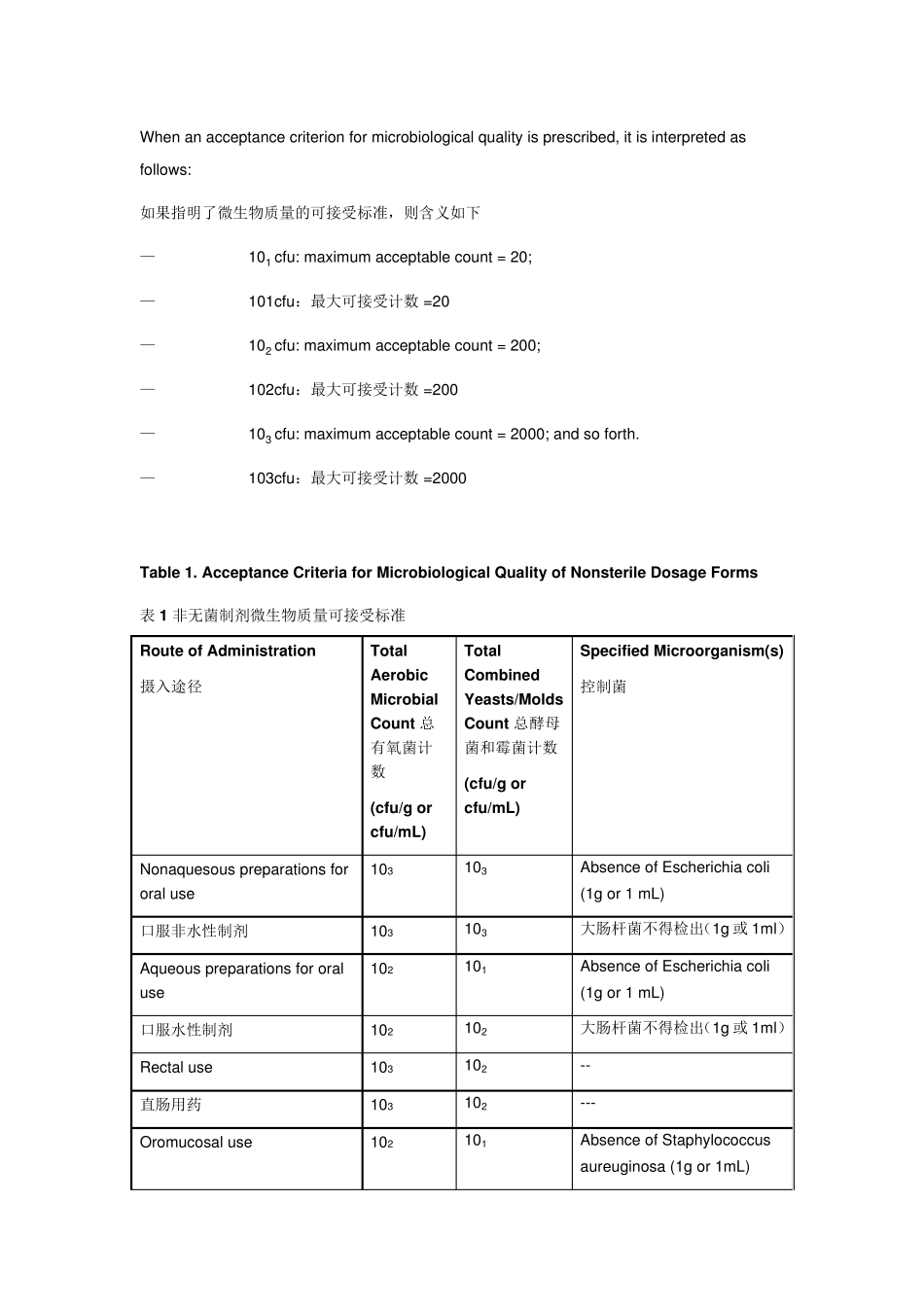
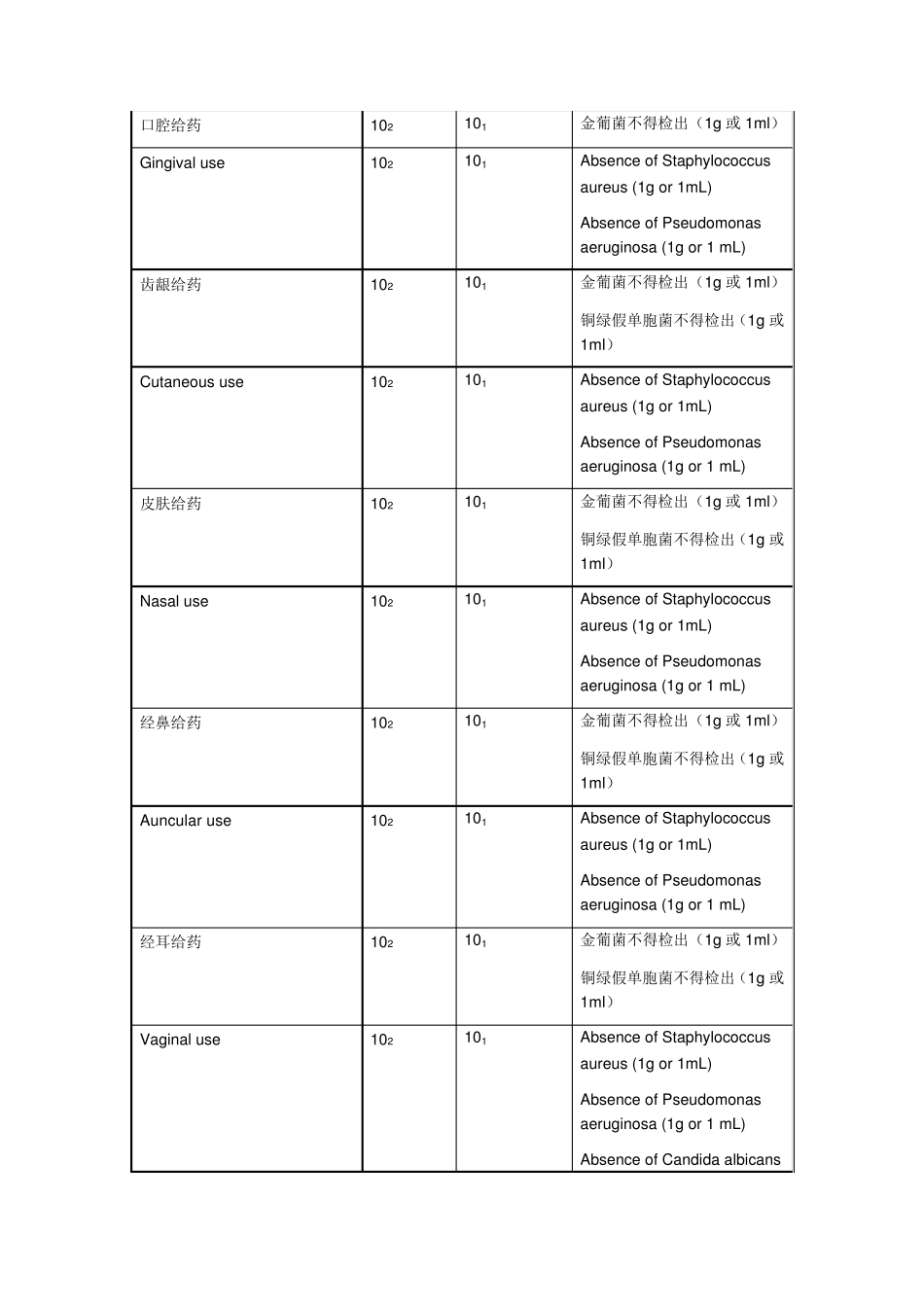
USP37 非 无 菌 药 品 微 生 物 检 查 :药 用 制 剂 和 药 用 物 质 可 接 受 标准 ( 中英文) (2014-06-06 14:19:33) 转载▼ 分类: 食品药品 MICROBIOLOGICAL EXAMINATION OF NONSTERILE PRODUCTS: ACCEPTANCE CRITERIA FOR PHARMACEUTICAL PREPARATIONS AND SUBSTANCES FOR PHARMACEUTICAL USE 非无菌药品微生物检查:药用制剂和药用物质可接受标准 The presence of certain microorganisms in nonsterile preparations may have the potential to reduce or even inactivate the therapeutic activity of the product and has a potential to adversely affect the health of the patient
Manufacturers have therefore to ensure a low bioburden of finished dosage forms by implementing current guidelines on Good Manufacturing Practice during the manufacture, storage, and distribution of pharmaceutical preparations
在非无菌制剂中如果出现特定的微生物,可能会降低甚至失去治疗活性,对患者健康产生潜在负面影响
因此,生产商需要通过在生产、存贮制剂的过程中实施GMP 现行指南,保证制剂