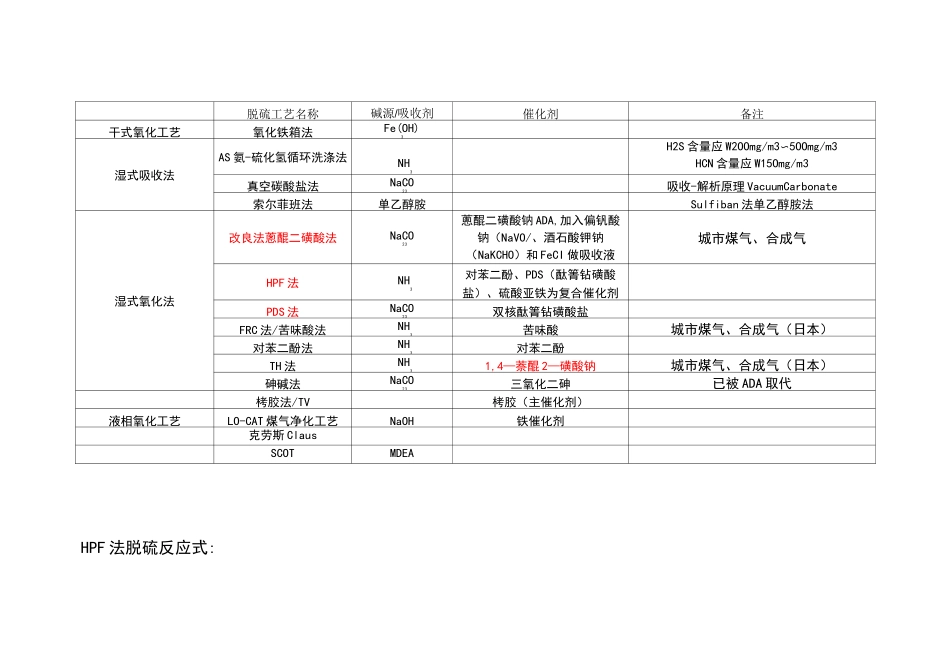
脱硫工艺名称碱源/吸收剂催化剂备注干式氧化工艺氧化铁箱法Fe(OH)3湿式吸收法AS氨-硫化氢循环洗涤法NH3H2S含量应W2OOmg/m3〜5OOmg/m3HCN含量应W15Omg/m3真空碳酸盐法NaCO23吸收-解析原理VacuumCarbonate索尔菲班法单乙醇胺Sulfiban法单乙醇胺法湿式氧化法改良法蔥醌二磺酸法NaCO23蔥醌二磺酸钠ADA,加入偏钒酸钠(NaVO/、酒石酸钾钠(NaKCHO)和FeCI做吸收液城市煤气、合成气HPF法NH3对苯二酚、PDS(酞箐钻磺酸盐)、硫酸亚铁为复合催化剂PDS法NaCO23双核酞箐钻磺酸盐FRC法/苦味酸法NH3苦味酸城市煤气、合成气(日本)对苯二酚法NH3对苯二酚TH法NH31,4—萘醌2—磺酸钠城市煤气、合成气(日本)砷碱法NaCO23三氧化二砷已被ADA取代栲胶法/TV栲胶(主催化剂)液相氧化工艺LO-CAT煤气净化工艺NaOH铁催化剂克劳斯ClausSCOTMDEAHPF法脱硫反应式:(1)脱硫反应:NH+HO=NHOH324NHOH+HS=NHHS+H042422NHOH+HS=(NH)S+2H042422NHOH+HCN=NHCN+H0442NHOH+CO=NHHCO4243NHOH+NHHCO二(NH)CO+H04434232NHOH+NHS+(x-1)S=(NH)S+H04442x22NHHS+(NH)CO+2(x-1)S=2(NH)S+CO+H0442342x22NHHS+NHHCO+(x-1)S=(NH)Sx+CO+H04434222NHCN+(NH)Sx=NHCNS+(NH)S442442(x-1)(NH)S+S=(NH)S42(x-1)42x(2)再生反应:NHHS+1/20二SJ+NHOH424(NH)S+1/2O+HO=SJ+2NHOH42224(NH)S+1/2O+HO=SJ+2NHOH