北京协和建昊医药技术开发有限责任公司Beijing Uuion-Genius Pharmaceutical Technology Ltd 项目领域Testing field序号No
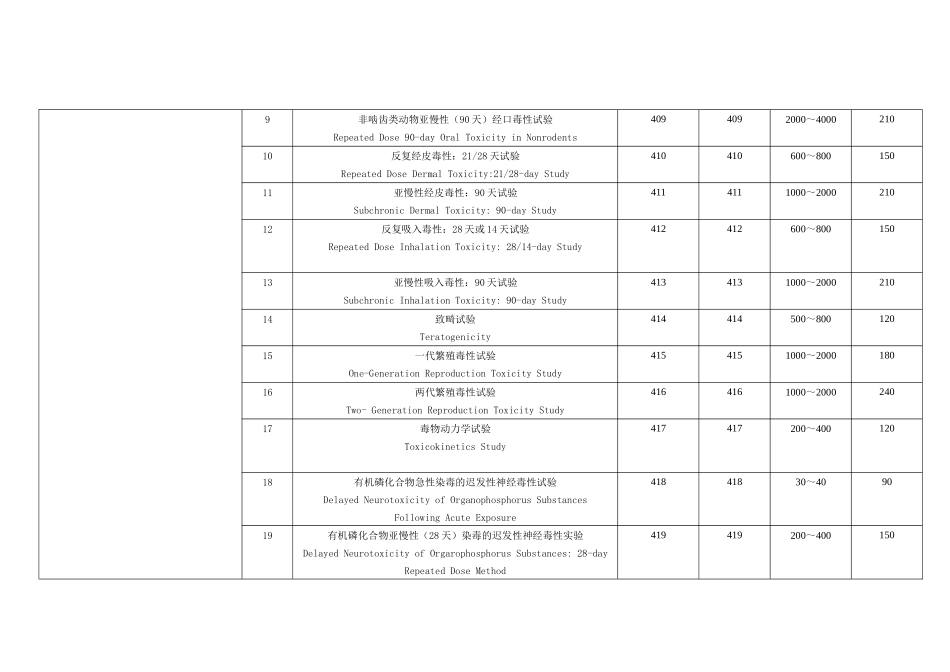
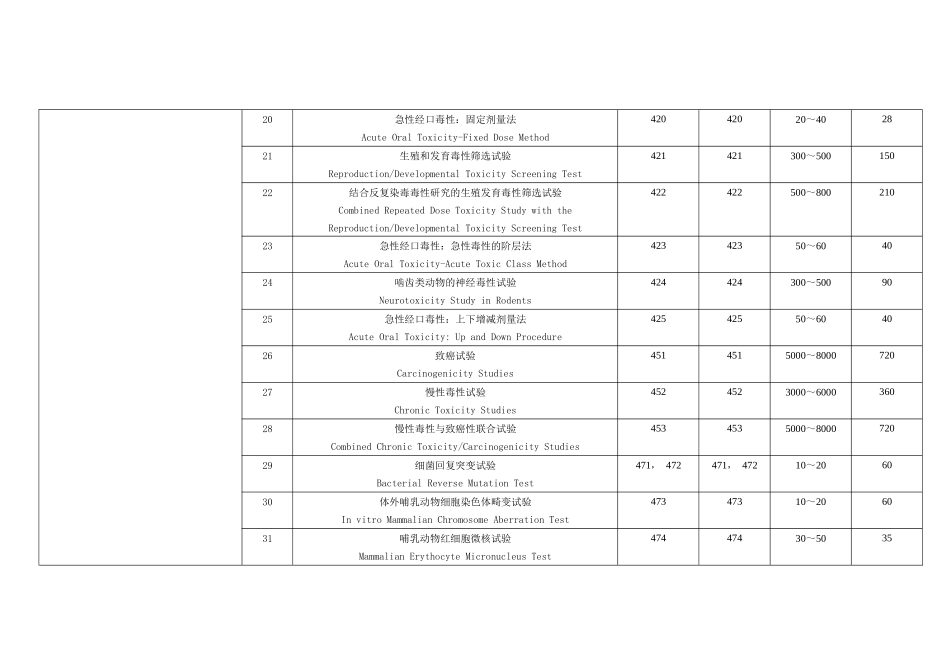
项目名称Name of study方法号Method codeOECD 编号OECD code所需样品量(克)Needed sample(g)所需时间(天)Needed Time(d)健康毒理Health Effects1急性经口毒性试验Acute Oral Toxicity Test40140120~30282急性经皮毒性试验Acute Dermal Toxicity Test40240210~20283急性吸入毒性试验Acute Inhalation Toxicity Test40340320~30284急性皮肤刺激性/腐蚀性试验Acute Dermal Irritation/Corrosion Test40440410~20285急性眼刺激性/腐蚀性试验Acute Eye Irritation/Corrosion Test40540510~20286皮肤致敏试验Skin Sensitization Test40640620~30407啮齿类动物 28 天反复经口毒性试验Repeated Dose 28-day Oral Toxicity Study in Rodents407407600~8001508啮齿类动物亚慢性(90 天)经口毒性试验Repeated Dose 90-day Oral Toxicity in Rodents4084081000~20002109非啮齿类动物亚慢性(90 天)经口毒性试验Repeated Dose 90-day Oral Toxicity in Nonrodents4094092000~400021010反复经皮毒性:21/28 天试验Repeated