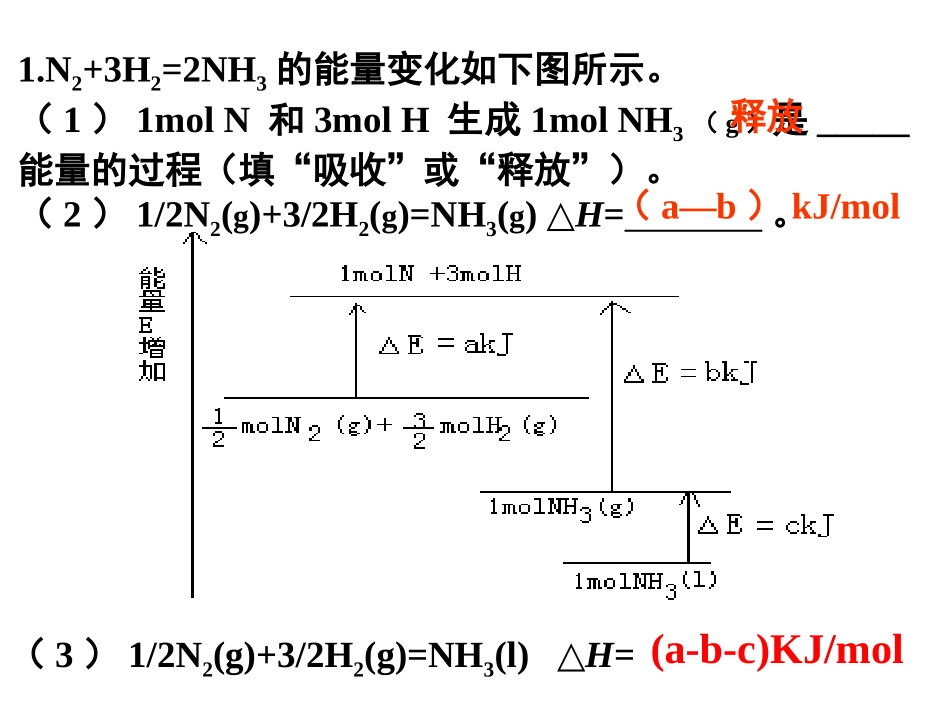
N2+3H2=2NH3的能量变化如下图所示
(1)1molN和3molH生成1molNH3(g)是_____能量的过程(填“吸收”或“释放”)
(2)1/2N2(g)+3/2H2(g)=NH3(g)△H=
释放(a—b)kJ/mol(3)1/2N2(g)+3/2H2(g)=NH3(l)△H=(a-b-c)KJ/mol11
化学反应N2+3H2=2NH3的能量变化如图所示,该反应的热化学方程式是()A.N2(g)+3H2(g)=2NH3(1)△H=2(a-b-c)kJ·mol-1B.N2(g)+3H2(g)=2NH3(g)△H=2(b-a)kJ·mol-11232N2(g)+H2(g)=NH3(g)△H=(a+b)kJ·mol-1D.1232N2(g)+H2(g)=NH3(1)△H=(b+c-a)kJ·mol-1C.Aa-b2(a-b)a-b-c一般指:C--CO2(g)S–SO2(g)H2O–H2O(l)定义:25℃,101kPa时,1mol纯物质完全燃烧生成稳定的化合物时所放出的热量
一、燃烧热:CH4(g)+2O2(g)=CO2(g)+2H2O(l)H=-890
3kJ/mol△分析以下几个热化学方程式,哪个是表示碳和氢气燃烧时的燃烧热的
C(s)+O2(g)==CO(g)ΔH=-110
5kJ/molB
C(s)+O2(g)==CO2(g)ΔH=-393
5kJ/molC
2H2(g)+O2(g)===2H2O(l)ΔH=-571
6kJ/molD
H2(g)+1/2O2(g)===H2O(g)ΔH=-241
8kJ/mol下列各组物质的燃烧热相等的是:A
碳和一氧化碳B
1mol碳和2mol碳C
1mol乙烯和2mol碳D
淀粉和纤维素H2(g)+1/2O2(g)=H2O(l)=-285
8kJ/molHH2(g)+1/2O2(g)=H2O(g)=-