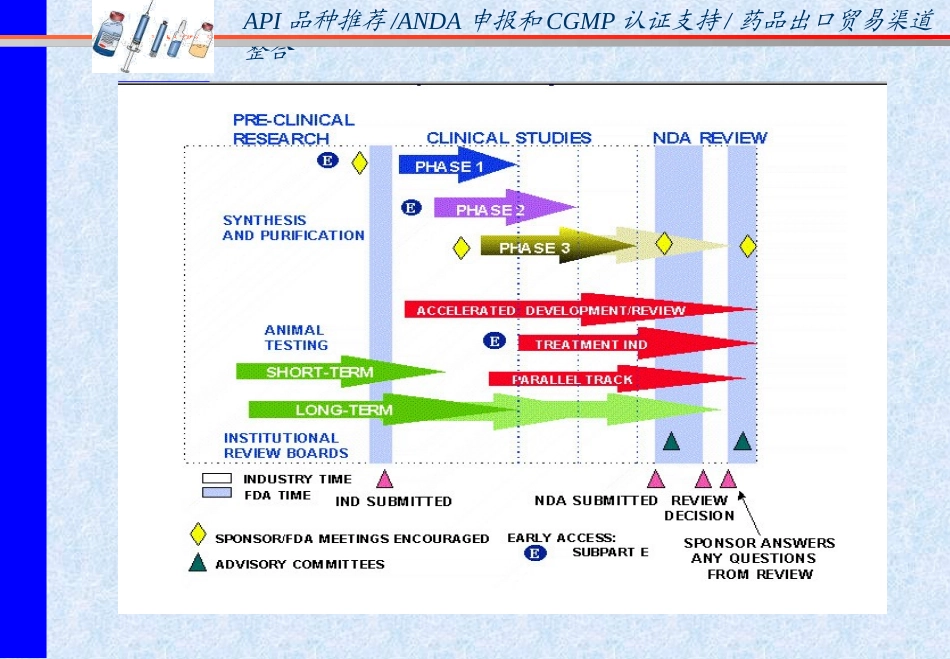
API品种推荐/ANDA申报和CGMP认证支持/药品出口贸易渠道整合FDADrugApprovalProcessFDA药品批准程序fromChemistryManufacturingControlsPerspective化学生产控制(CMC)展望上海宝钜API品种推荐/ANDA申报和CGMP认证支持/药品出口贸易渠道整合NewDrugDevelopmentProcess新药的研发过程1
Research&Development(研究和开发)2
Pre-clinicalStudies(药理毒理研究)3
InvestigationalNewDrug(IND)PhaseI,II,IIIClinicalTrials(临床试验)4
NewDrugApplication(NDA)(新药申请)5
Post-marketing(新药批准后的市场调查跟踪)Post-approvalchanges(批准后的生产工艺变更)API品种推荐/ANDA申报和CGMP认证支持/药品出口贸易渠道整合8API品种推荐/ANDA申报和CGMP认证支持/药品出口贸易渠道整合NewDrugDevelopmentProcess新药研发程序NewChemicalEntity-OrganicSynthesis-NaturalProductPre-clinicalStudies-Physi/chemProperties-Biologicalactivities-Pre-formulationInvestigationalNewDrugApplicationClinicalTrials-PhaseI,II,IIIStudies:-LongtermAnimalToxicity-ProductFormulationDevelopment-ManufacturingandControls-PackageandLabelDesig