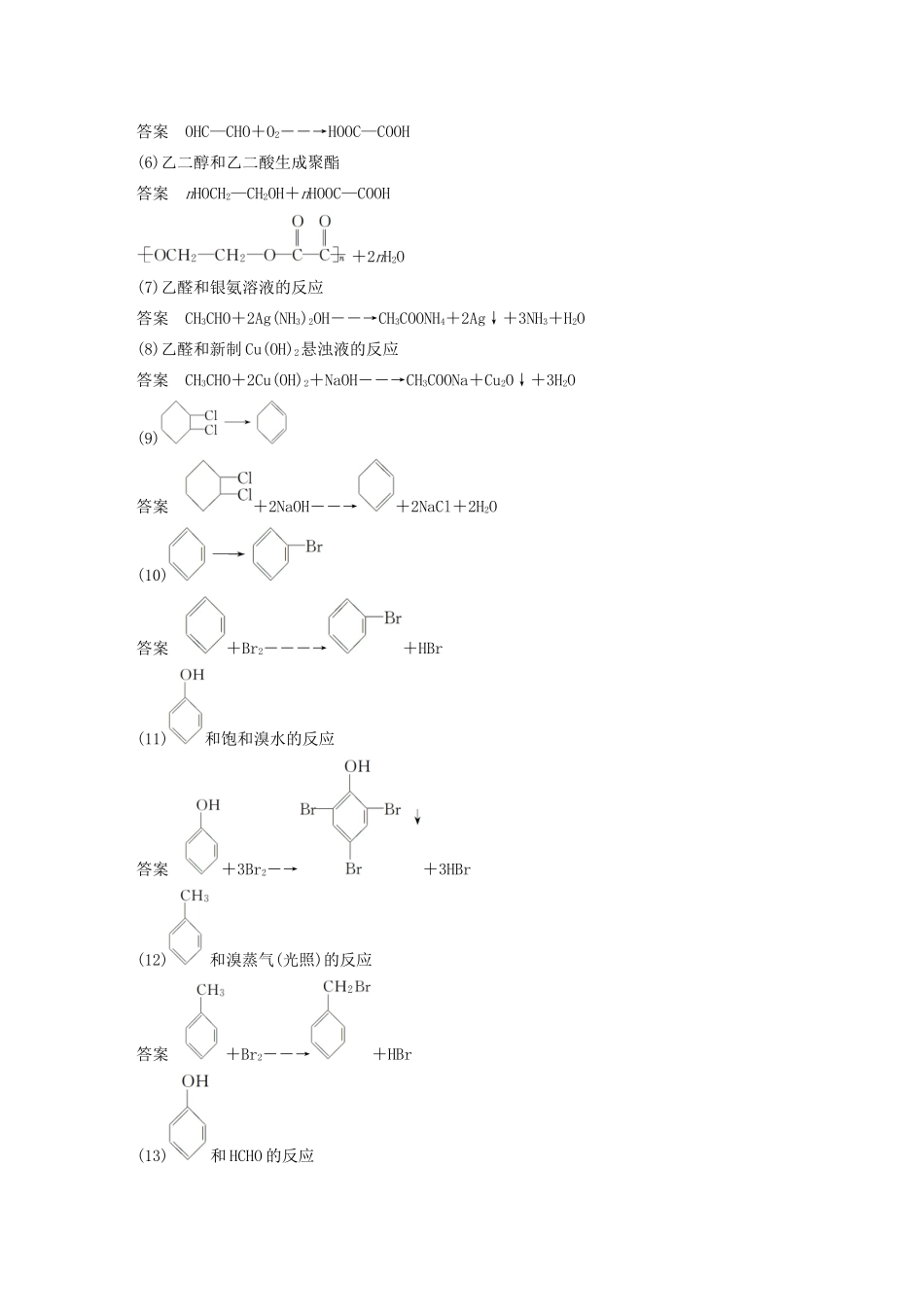
本章重要有机物之间的转化关系一、重要有机物之间的转化关系二、重要反应必背(1)CH3CH2Cl―→CH2==CH2答案CH3CH2Cl+NaOH――→CH2==CH2↑+NaCl+H2O(2)CH3CH2OH―→CH2==CH2答案CH3CH2OH――――→CH2==CH2↑+H2O(3)CH3CH2OH―→CH3CHO答案2CH3CH2OH+O2――→2CH3CHO+2H2O(4)CH3CH2OH和CH3COOH生成乙酸乙酯答案CH3COOH+CH3CH2OHCH3COOC2H5+H2O(5)OHC—CHO―→HOOC—COOH答案OHC—CHO+O2――→HOOC—COOH(6)乙二醇和乙二酸生成聚酯答案nHOCH2—CH2OH+nHOOC—COOH+2nH2O(7)乙醛和银氨溶液的反应答案CH3CHO+2Ag(NH3)2OH――→CH3COONH4+2Ag↓+3NH3+H2O(8)乙醛和新制Cu(OH)2悬浊液的反应答案CH3CHO+2Cu(OH)2+NaOH――→CH3COONa+Cu2O↓+3H2O(9)答案+2NaOH――→+2NaCl+2H2O(10)答案+Br2―――→+HBr(11)和饱和溴水的反应答案+3Br2―→+3HBr(12)和溴蒸气(光照)的反应答案+Br2――→+HBr(13)和HCHO的反应答案+nHCHO――→+nH2O(14)酯在碱性条件的水解(以乙酸乙酯在NaOH溶液中为例)答案CH3COOC2H5+NaOH――→CH3COONa+C2H5OH(15)和NaOH的反应答案+2NaOH――→CH3COONa++H2O三、有机物的检验辨析1.卤代烃中卤素的检验取样,滴入NaOH溶液,加热至分层现象消失,冷却后加入稀硝酸酸化,再滴入AgNO3溶液,观察沉淀的颜色,确定是何种卤素
2.烯醛中碳碳双键的检验(1)若是纯净的液态样