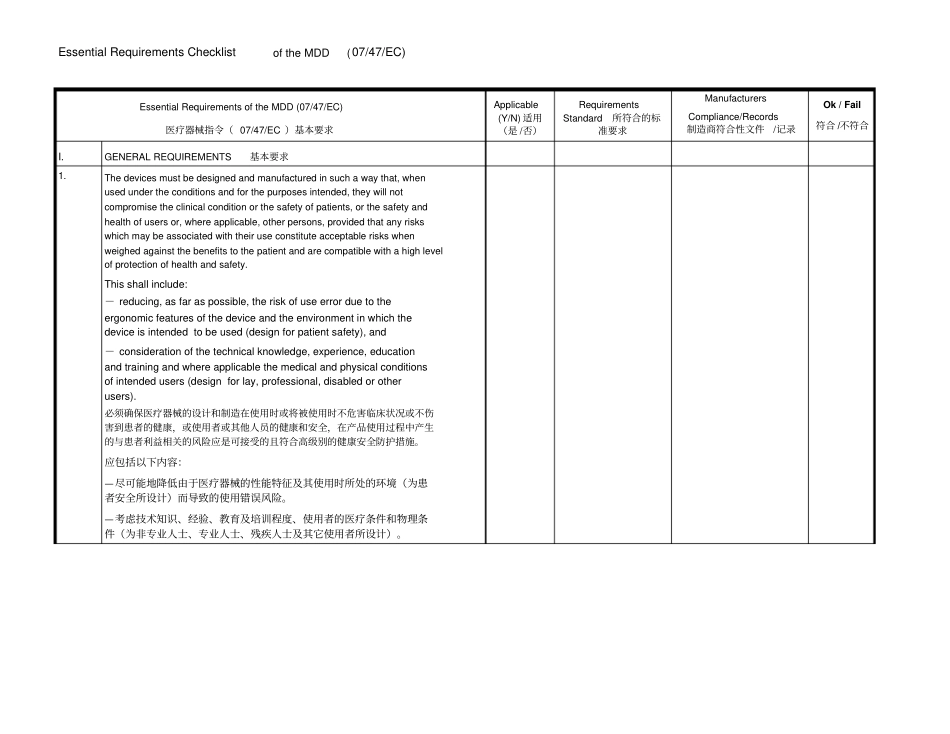
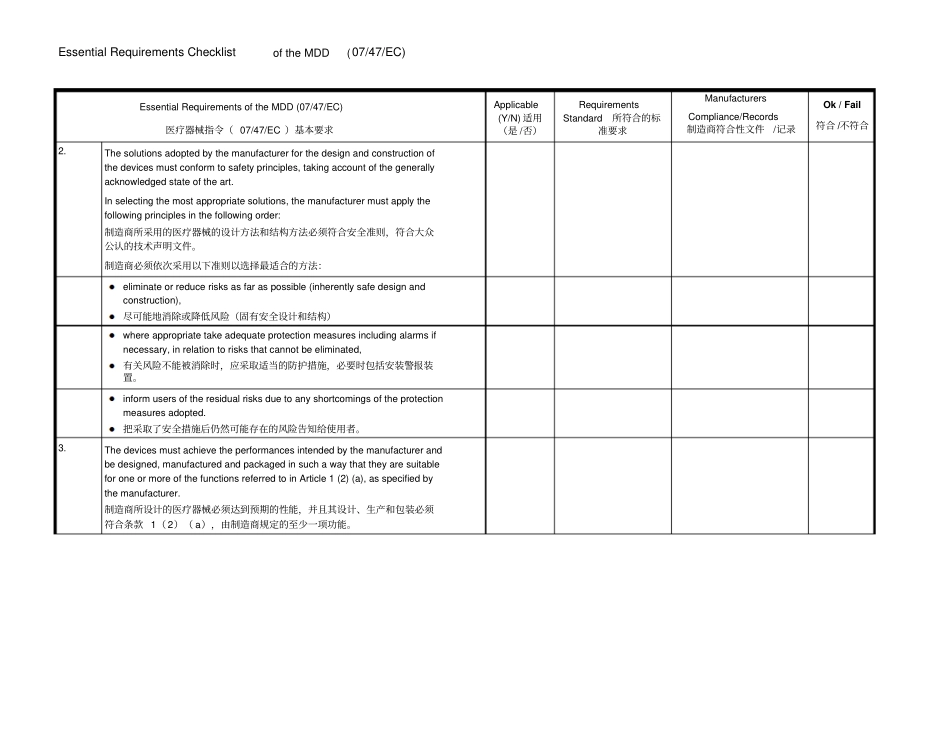
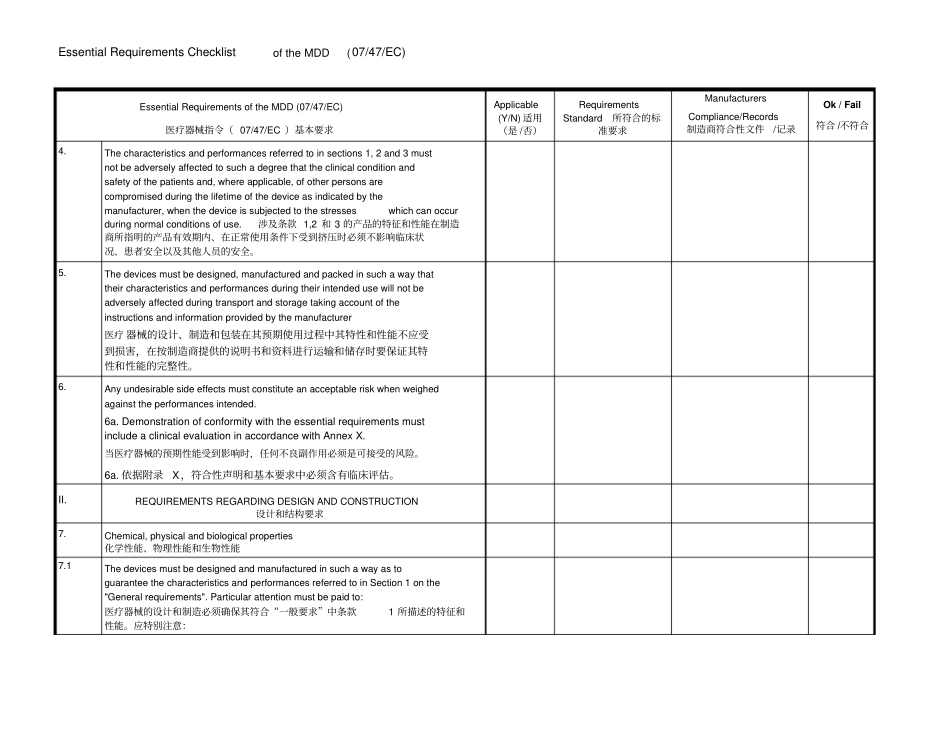
Essential Requirements Checklist of the MDD (07/47/EC) Essential Requirements of the MDD (07/47/EC) 医疗器械指令( 07/47/EC )基本要求Applicable (Y/N) 适用(是 /否)Requirements Standard 所符合的标准要求Manufacturers Compliance/Records 制造商符合性文件/记录Ok / Fail 符合 /不符合I
GENERAL REQUIREMENTS基本要求1
The devices must be designed and manufactured in such a way that, when used under the conditions and for the purposes intended, they will not compromise the clinical condition or the safety of patients, or the safety and health of users or, where applicable, other persons, provided that any risks which may be associated with their use constitute acceptable risks when weighed against the benefits to the patient and are compatible with a high level of protection of health and safety
This shall include:- reducing, as far as possib